Consumer Trials vs. Clinical Trials
What's the Difference, and Which Do You Need?
CONSUMER HEALTH STUDIES
4/13/20264 min read


If you've been researching how to validate your supplement, skincare, or wellness product, you've probably run into both terms: consumer trial and clinical trial. They sound similar. They both involve testing a product on people. But they are fundamentally different in purpose, cost, timeline, and what they can do for your brand.
Understanding the difference between a consumer trial vs clinical trial for your supplement or wellness product could save you years of time and tens of thousands in budget, while still getting you the evidence you actually need.
Let's break it down.
What Is a Clinical Trial?
A clinical trial is a rigorously controlled scientific study, typically governed by regulatory bodies, designed to test whether a substance (usually a drug or medical intervention) produces a specific physiological effect in patients with a diagnosed condition.
Clinical trials exist to satisfy regulatory agencies, the FDA in the US, the EMA in Europe, and their equivalents globally. They follow strict protocols, require oversight from an Institutional Review Board (IRB), and are often registered on databases like ClinicalTrials.gov.
The key characteristics:
Participants: Usually patients with specific conditions, not healthy consumers
Oversight: Heavily regulated with IRB approval and regulatory filings
Controls: Typically randomised and double-blind (neither participants nor researchers know who receives the active product)
Goal: Prove a substance is safe and effective for regulatory drug approval
Timeline: Months to years (Phase I, II, III trials can span a decade)
Cost: Hundreds of thousands to tens of millions of euros
Clinical trials are built for the pharmaceutical industry. They're designed to answer the question: "Is this drug safe and effective enough to be approved for medical use?"
That is almost certainly not the question you need to answer.
What Is a Consumer Trial?
A consumer trial is a structured study in which real consumers, your target audience, use your product under defined conditions, and their experiences, outcomes, and perceptions are systematically measured.
Consumer trials are designed for the consumer health market: supplements, skincare, functional foods, and wellness products. They're built to answer the questions that matter for a brand:
Does this product produce measurable, noticeable results in real users?
What outcomes can we legitimately claim in our marketing?
How do consumers experience the product, efficacy, texture, tolerance, taste?
They're rigorous in methodology, but designed for consumer products, not pharmaceutical approval. That distinction matters enormously.
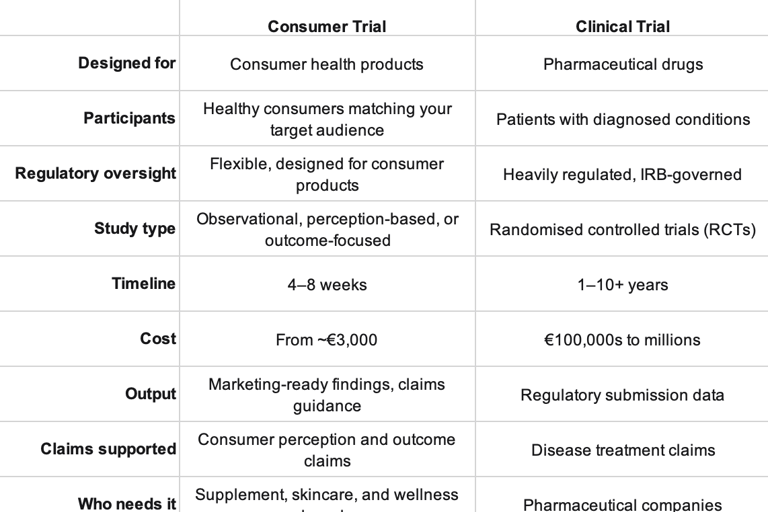
Side-by-Side: Consumer Trial vs. Clinical Trial
Consumer TrialClinical TrialDesigned forConsumer health productsPharmaceutical drugsParticipantsHealthy consumers matching your target audiencePatients with diagnosed conditionsRegulatory oversightFlexible, designed for consumer productsHeavily regulated, IRB-governedStudy typeObservational, perception-based, or outcome-focusedRandomised controlled trials (RCTs)Timeline4–8 weeks1–10+ yearsCostFrom ~€3,000€100,000s to millionsOutputMarketing-ready findings, claims guidanceRegulatory submission dataClaims supportedConsumer perception and outcome claimsDisease treatment claimsWho needs itSupplement, skincare, and wellness brandsPharmaceutical companies
The Confusion in the Market
Here's where many brands go wrong: they assume that because they can't do a clinical trial, they can't produce credible evidence at all. So they don't do anything, and end up with vague, unsubstantiated marketing claims that no one believes.
Or, they do an informal customer survey and call it a "study." That's the other extreme, and it creates legal and reputational risk when buyers, retailers, or regulators look closely.
The truth is there's a structured, credible middle ground: a properly designed consumer trial that gives you real data, usable claims, and a paper trail that supports your marketing.
The distinction matters for compliance, too. Consumer trials produce perception and outcome claims, things like "8 out of 10 participants noticed improved energy after 30 days", which are appropriate for supplement and wellness marketing. Clinical trial language (e.g., "clinically proven to treat...") is reserved for studies that meet pharmaceutical standards, and misusing it is a regulatory red flag in most markets.
Which One Do You Actually Need?
You need a consumer trial if:
You're a supplement, skincare, longevity, or wellness brand
You want to make efficacy or perception claims in your marketing
You're preparing for a retail launch or pitching to buyers
You want science-backed content that builds trust with your audience
You're working with a realistic budget and timeline (weeks, not years)
You might need a clinical trial if:
You are developing a pharmaceutical drug or medical device
You intend to make disease-treatment claims regulated by health authorities
You are seeking regulatory approval from the FDA, EMA, or similar bodies
You have the budget, team, and multi-year timeline to support the process
For the overwhelming majority of supplement and wellness brands, from solo founders launching their first product to established enterprises expanding a product line, a consumer trial is the right tool.
What Consumer Trial Results Actually Look Like
To make this concrete, here are the types of findings a well-run consumer trial can produce:
"87% of participants reported reduced bloating within 2 weeks of daily use"
"Participants showed a statistically significant improvement in self-reported sleep quality over 6 weeks"
"9 out of 10 participants said their skin felt more hydrated after 4 weeks"
"Average energy scores increased by 34% from baseline over the 30-day study period"
These are specific, credible, and directly usable in marketing copy, retailer presentations, press materials, and product packaging. They're not vague ingredient claims, they're outcome data from real users of your product.
A Note on Study Design
Whether you're looking at a consumer trial or something more involved, the quality of the study is everything. A study's credibility comes down to:
Clear objectives, what specific outcomes are being measured, and why?
Appropriate participant recruitment, are participants representative of your actual target customer?
Validated measurement tools, are outcomes being assessed using recognised, reproducible methods?
Statistical rigour, is the sample size large enough for the findings to be meaningful?
Transparent methodology, could another researcher replicate this study from the documentation?
This is why working with a specialist provider, rather than running an internal test, makes the difference between data you can stand behind and data you can't.
The Bottom Line
Clinical trials are built for drug approval. Consumer trials are built for brands.
If you're a supplement, skincare, or wellness brand that wants to validate your product, make credible marketing claims, and build long-term trust with customers and retailers, a consumer trial is what you need. It gives you the evidence without the pharmaceutical price tag, timeline, or regulatory burden.
The key is doing it properly: structured methodology, clear objectives, validated tools, and results you can actually use.
Want to find out what a consumer trial could look like for your product?
Book a free consultation with the Validence Labs team →
Looking to understand more about what consumer trials involve? Read our previous post: What Is a Consumer Trial? How Health Brands Validate Products Without Clinical Studies
Validence Labs
We specialize in consumer insight studies and biomarker-supported consumer research for supplements, skincare, wellness, and functional products. Based in Germany and serving clients across the world, we help brands validate claims, understand customers, and make data-driven decisions.
Contact
info@validencelabs.com
© 2025. All rights reserved.
